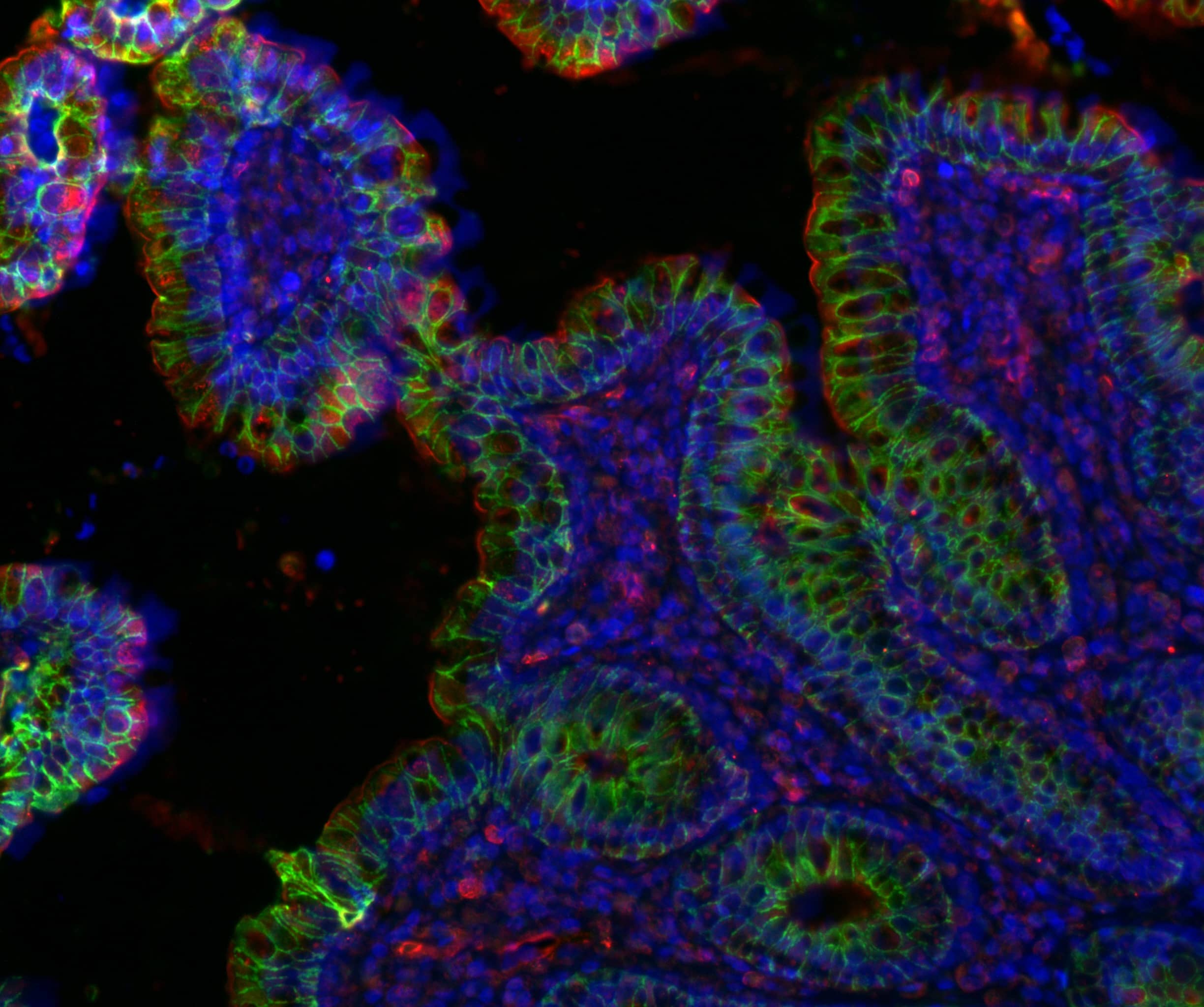
The mammalian intestine performs diverse and essential tasks such as digestion, nutrient absorption, and immunity. In the Kulkarni Gut Lab, we study how the intestinal barrier maintains homeostasis; how it responds to external stressors such as pathogens and toxins, and finally how it recovers after an injury. We have the following major research areas:
Epithelial Encounter with Luminal Matter
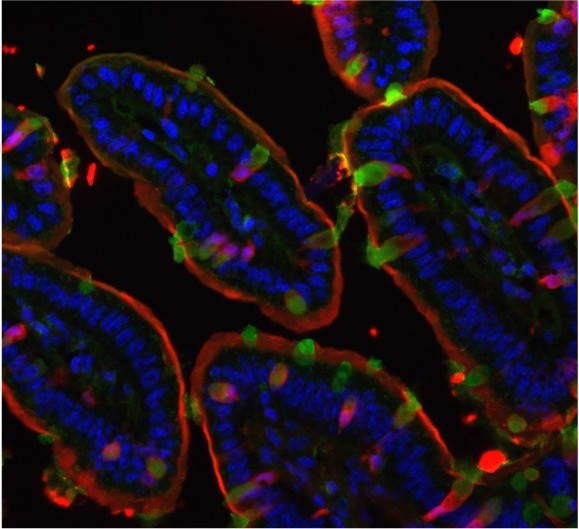
We have demonstrated that intestinal goblet cells and their ability to form goblet cell-associated antigen passages (GAPs) is a major pathway for steady-state luminal antigen transfer to lamina propria-antigen presenting cells (LP-APCs). Our data suggest that GAPs are necessary for 1) the imprinting of APCs with tolerogenic properties, 2) the de novo induction of Tregs specific for dietary antigens, and 3) the maintenance of pre-existing LP iTregs (Kulkarni et. al Mucosal Immunology 2020). Using biopsy specimens, we have observed robust number of GAPs in human tissue, and identified potential triggers that alter this pathway. Our goal is to dissect how this pathway helps maintain intestinal homeostasis and how immune balance is altered when GAPs are modulated.
Novel Role of Innate Immune Proteins
We and others have demonstrated the activity of complement components such as C3 and Factor B (CFB) at mucosal sites such as the gut, lungs, genitourinary tract and skin, which are exposed to various innocuous and at times deleterious antigens. Yet, a comprehensive understanding of cell type-specific sourcing of complement proteins, and their activation and regulation at mucosal sites, especially the gut, remains unknown. A major hurdle for investigating the tissue-specific roles of complement has been the limited availability of models and assays. We have developed key transgenic models and functional assays to separate the roles of circulating versus gut-derived complement proteins, which will help us delineate the role of complement in intestinal immunity. Our overarching theme is that at intestinal barrier site, the high likelihood of external threats requires local rather than systemic complement activation and regulation to orchestrate homeostasis.